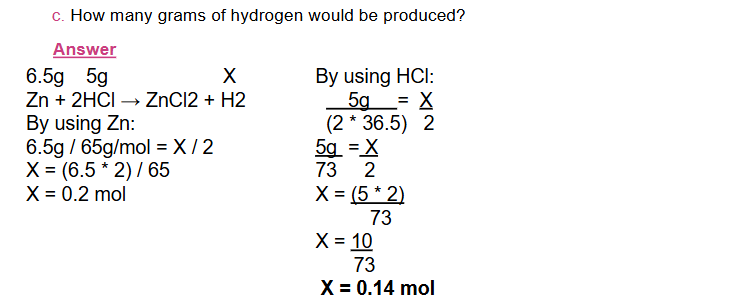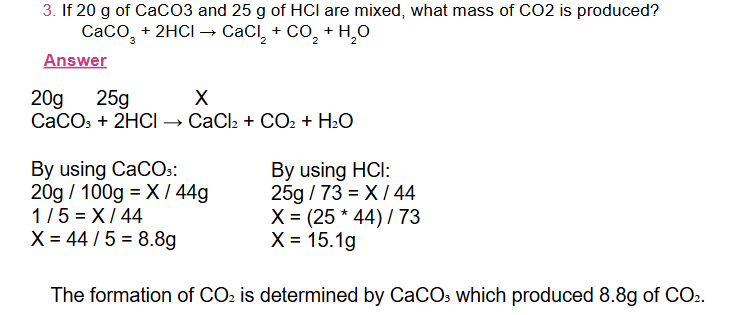-
Stoichiometry is the study of the quantitative composition of substances and the relationship that exists between the reactants and the products involved in chemical reactions.
-
It is the study of the amount or ratio of moles, mass, or volumes (for gases) of reactants and products.
-
Stoichiometric calculations are based on the following two major principles.
-
The composition of any substance in the chemical equation should be expressed by a definite formula.
-
The law of conservation of mass must be obeyed (the mass of reactants equals the mass of products)
Note That stoichiometric calculations are done for balanced chemical equations.

-
Stoichiometry problems of this type are called either mass-mass or mole-mole problems.
-
A balanced chemical equation provides information about the nature, masses, number of moles, number of molecules/atoms of reactants and products
-
For example, consider the reaction of hydrogen with nitrogen to produce ammonia.
-
molecules of hydrogen and 1 molecule of N2 react to produce 2 molecules of ammonia.
-
The reaction of 3 moles (3x6.02 x 1023 molecules) of H2 with 1 mole (6.02 X 1023 molecules) of N2 to yield 2 moles (2 x 6.02 x 1023 molecules ) of NH3.
-
The same equation can also tell us the combination of 6 g of H2 with 28 g of N2 to yield 34 g of NH3. This can be further interpreted as follows
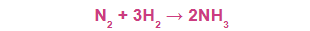
-
1 molecule N2 + 3 molecules H2 → 2 molecules NH3 (Molecular interpretation)
-
1 mol N2 + 3 mol H2 → 2 mol NH3 (Molar interpretation)
-
28.0 g N2 + 3 x 2.02 g H2 → 2 x 17.0 g NH3 ( mass interpretation)
-
Calculations based on chemical equations (stoichiometric problems) are classified into
-
Mass-mass,
-
volume-volume and
-
mass-volume problems

-
There are two methods for solving mass-mass problems.
-
These are mass-ratio and mole-ratio methods.
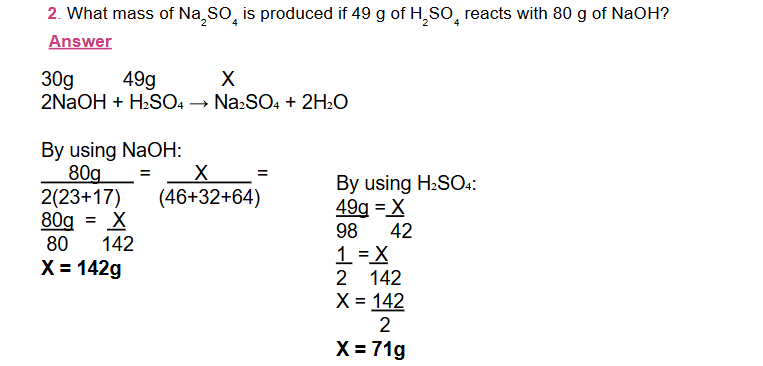
A. The Mass - ratio Method
-
In this method, the mass of one substance is determined from the given mass of the other substance using the following steps.
Step 1: Write the balanced chemical equation.
Step 2: Place the given mass above the corresponding formula, and x above the formula of the substance whose mass is to be determined
Step 3: Write the total molar mass of the substances below the formula of each substance.
(Total molar mass is the molar mass of the substance multiplied by its coefficient).
Step 4: Set up the proportion.
Step 5: Solve for the unknown mass, x.
Example: How many grams of aluminum metal must be heated to produce 20.4 g of aluminium oxide?
Note that stoichiometric problems can also be solved using the mole ratio or molar mass as conversion factors.
-
The numbers in a conversion factor come from the coefficients of the balanced chemical equation
-
In general, for mole – mole problems,
mole A x mole ratio = mole B

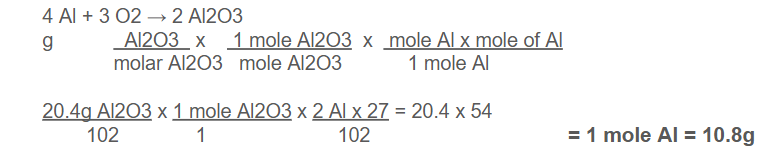
-
In the above example, coefficients of Al and Al₂O₃ are 4 and 2 respectively
-
Therefore, conversion factor = mole ratio = 4 mol Al ∕2 mol Al2O3. Given molar masses,
1 mole Al₂O₃= 102 g, 1 mole Al = 27 g
-
The conversion steps are summarized as follows:
grams of Al2O3 → moles of Al2O3→ moles of Al → grams of Al
Conversion factor: molar mass of Al2O3 mole ratio molar mass of Al
20.4 g x 1 mol Al2O3 /102 g Al2O3x 4 mol Al /2 mol Al2O3x 27 g Al/1mol Al = 10.8 g
Example: What mass of magnesium sulfate will be produced if 4.8 g of magnesium reacts with excess sulfuric acid?

B. The Mole - ratio Method
-
In this method, calculations are made in terms of moles; therefore the given mass is converted into mole.
-
The obtained mole can be converted back to mass if required.
-
Follow the steps given below to solve problems of mass-mass relationships using mole ratio method:
Step 1: Write the balanced chemical equation.
Step 2: Convert the given mass to moles and write the obtained moles and the required quantity, x, above the formulas of the respective substances.
Step 3: Place the coefficients as the number of moles under the formula of each substance involved.
Step 4: Set up the proportion.
Step 5: Solve for the unknown value, x; and convert the moles obtained into mass.
Example 1.28:
How many grams of calcium oxide are needed to react completely with 22.0 g of carbon dioxide?
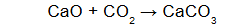

Example: How many grams of carbon monoxide must react with excess iron oxide to produce 28 grams of iron?
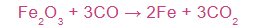
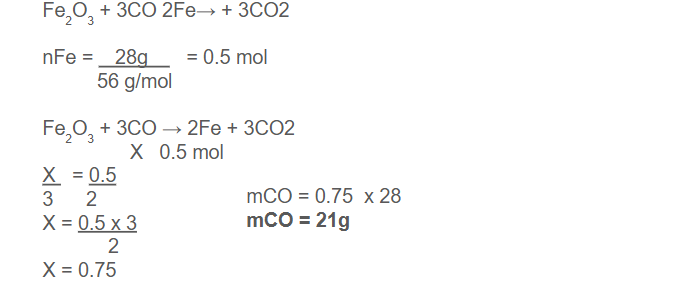
-
In mole-mass problems, the amount of one substance is given in moles and the mass of another substance is determined, usually in grams and in mass-mole problems, the mass of one substance is given, usually in grams and the amount of another substance is determined in moles.
-
for a mole-mass calculation follow the following sequence:
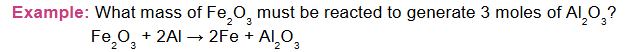
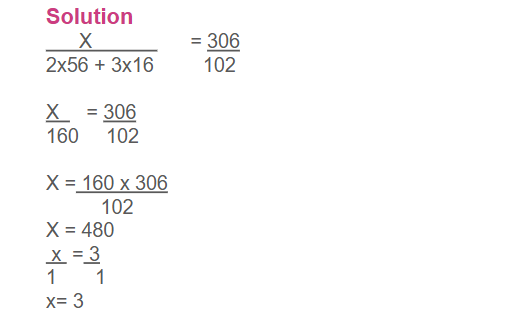
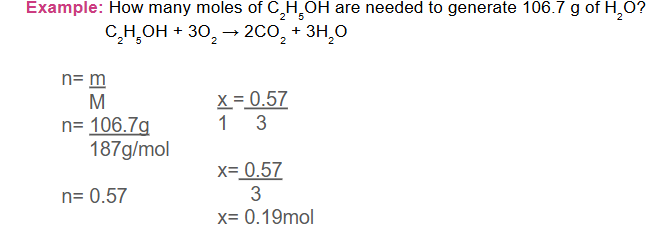





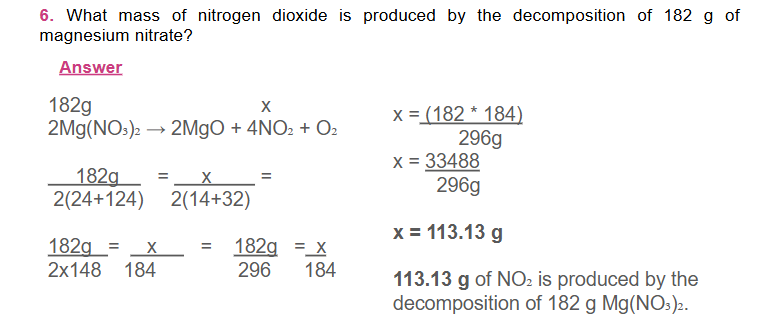

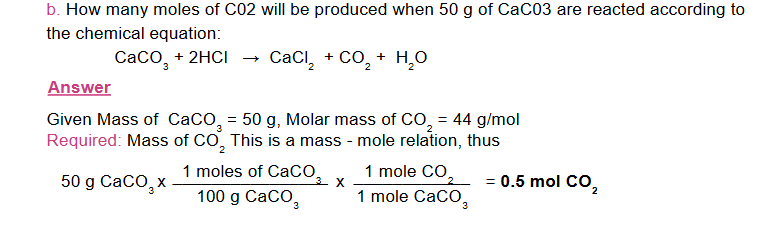
-
At Standard Temperature (00C) and Pressure (1 atm) or STP, one mole of any gas occupies 22.4 liters.
-
This, 22.4 liters is known as molar volume at STP.
-
The volume of a gas and its number of molecules are related and explained by Avogadro’s Law.
-
Avogadro’s Law states that equal volumes of different gases, under the same conditions of temperature and pressure, contain equal number of molecules.
-
According to this law, the volume of a gas is proportional to the number of molecules (moles) of the gas at STP as shown below:
V ~ n; where V is the volume and n is the number of moles gas
In Volume – volume problems, the volume of one substance is given and the volume of the other substance is calculated.




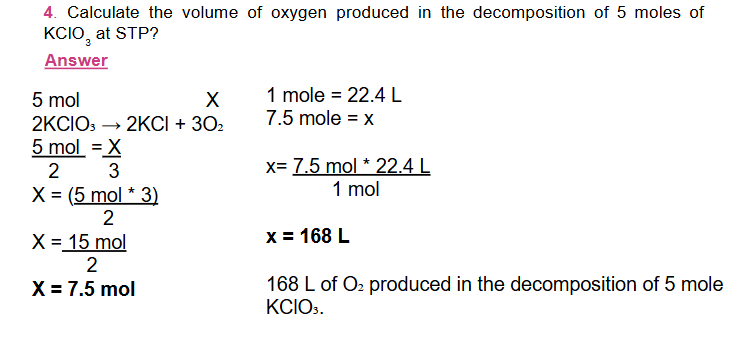


-
In mass-volume problems, either the mass of one substance is given and the volume of the other is required or the volume of one substance is given and the mass of the other one is required.
-
The steps to solve such type of problems are the same as the previous steps except putting the masses on one side and the volumes on the other side of the equality sign.






-
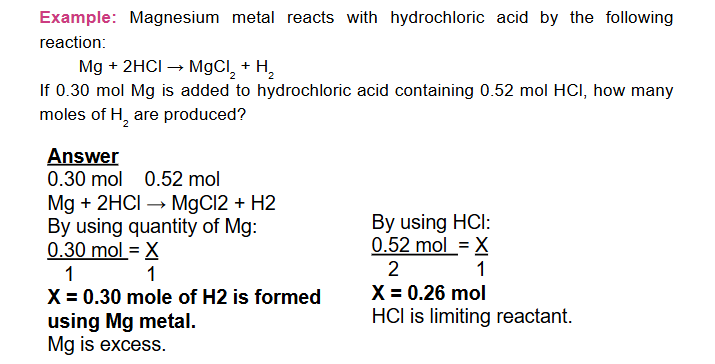
In a chemical reaction involving two reactants, the reaction will stop when all of one reactant has been completely consumed
-
The reactant completely consumed first in a reaction is called the limiting reactant, because it limits or determines the amount of product that can be formed.
-
When the limiting reactant is completely consumed, no more product can be formed and thus the other reactant remains excess.
-
Excess reactants are the reactants present in quantities greater than necessary to react with the quantity of the limiting reactant.
-
Suppose you want to make some cheese sandwiches. Each is made from two slices of bread and a slice of cheese
2 slices bread + 1 slice cheese → 1 cheese sandwich
If you have 7 slices of bread and 2 slices cheese, you can make 2 cheese sandwiches.
-
3 slices of bread is excess.
-
The number of sandwiches is limited by the number of slices of cheese.
-
Hence, slices of cheese are the limiting ingredient and slices of bread are excess ingredient.
Note that: Yield of the product is the one calculated using the limiting reactant and it is the smallest.
-