

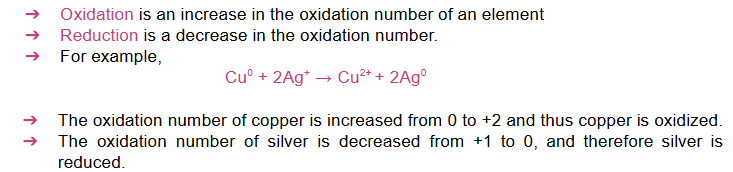











-
Whenever one substance loses an electron (is oxidized), another substance must gain that electron (be reduced).
-
The substance that gives up an electron and causes reduction is called a reducing agent.
-
The substance that gains an electron and causes the oxidation is called an oxidizing agent.

-
Tests for an oxidizing agent are accomplished by mixing it with a substance that is easily oxidized to give a visible color change when the reaction takes place.

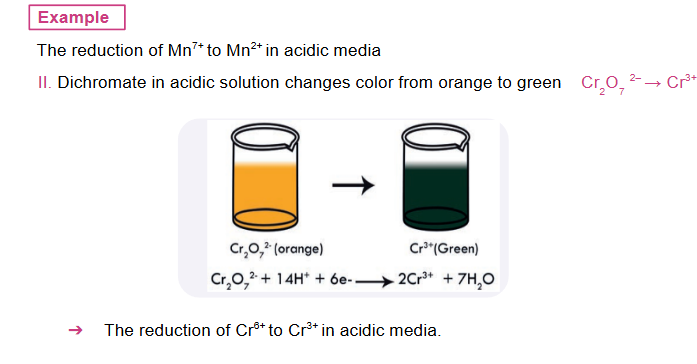





-
Oxidation and reduction or redox reactions occur simultaneously in a given reaction
Example 1.14: Identify the reducing and oxidizing agents in the following
balanced redox reactions:
2Fe + 3O₂ → Fe₂O₃
Assign oxidation states of the reactants and products, then identify the species oxidized and reduced.
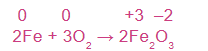
-
Fe is oxidized from 0 to +3
-
O is reduced from 0 to –2
Therefore, Fe is reducing agent and O₂ is oxidizing agent.

-
Mg is oxidized from 0 to +2, therefore Mg is reducing agent.
-
H is reduced from +1 to 0, therefore HCl is the oxidizing agent.

-
Where do the most easily reduced and oxidized elements found in the periodic table of the elements?

-
Transition elements at the right of the periodic table such as silver and copper can easily be reduced (strong oxidizing agents) from their compounds.
-
Alkali metals like potassium can easily be oxidized (strong reducing agents)















